Ice Cream Special
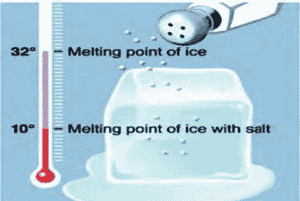
Students experience the importance of lowering the melting point of ice via the addition of salt, as well as the effect of continuously mixing ingredients to expose them to the coldest temperatures, as they make home-made ice cream.
Time
Materials
Instructions
Guiding Questions
STEM Connections
Enjoy this activity?
Join our community and receive activities, early access to programs, and other special news, and/or leave feedback on this activity below!
Submitting...
DiscoverE helps create activities like this around Forces, Motion & Energy, Mixtures & Solutions for children in Kindergarten to 2nd Grade and Elementary School and Middle School to help prepare them for STEM careers in fields like Agriculture & Biosystems or Chemical or Industrial & Manufacturing.

 Students experience the importance of lowering the melting point of ice via the addition of salt, as well as the effect of continuously mixing ingredients to expose them to the coldest temperatures, as they make homemade ice cream.
Students experience the importance of lowering the melting point of ice via the addition of salt, as well as the effect of continuously mixing ingredients to expose them to the coldest temperatures, as they make homemade ice cream.




Thank you! Your submission is processing.