This article contains collapsible sections with activity materials, instructions, and additional resources. Use the expand/collapse buttons to view each section.
Activity Content Sections
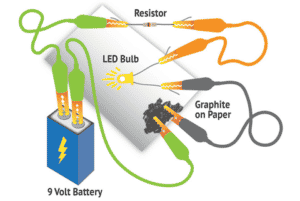
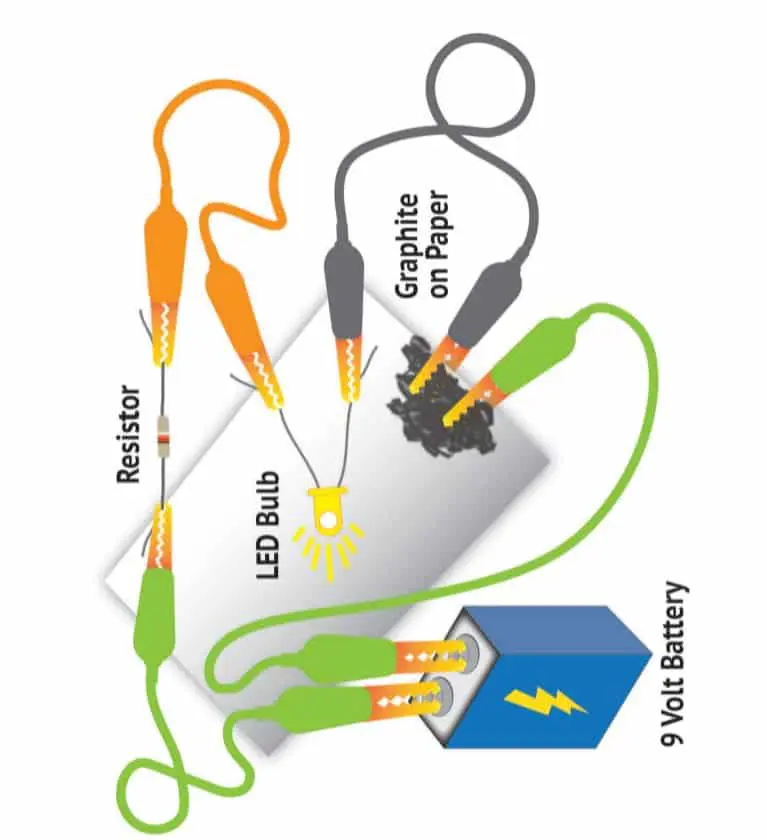
Click to expand or collapse the materials needed section










Leave feedback form
Thank you! Your submission is processing.